NanoAnalyzer
- Laser (blue, green, red)
- One SSC & Two FL channels
- Lower detection limit
- High detection limit (1 micron)
- Up to 12,000 particles/min
Single-Molecule NanoAnalyzer
- High-throughput
- Automatic clean-up
- Up to 3 markers
- New Features
Fluorescent quantitation
Nucleic acid copy number per LNP
Copy number of specific proteins
Encapsulation efficiency
Reagents Consumables
- Quality Control
- Size Standard
- Fluorescent quantitation
- Fluorescent Antibody

























EXTRACELLULAR VESICLES

THE ORIGIN AND PHENOTYPING OF EXTRACELLULAR VESICLES

VACCINE

COMPREHENSIVE MEASUREMENT SUITE FOR NANOSIZED DRUG PRODUCTS

NANOMEDICINE

COMPREHENSIVE MEASUREMENT SUITE FOR NANOSIZED DRUG PRODUCTS

BACTERIA DETECTION

TRULY QUANTITATIVE MEASUREMENT OF TOTAL BACTERIAL TITRE
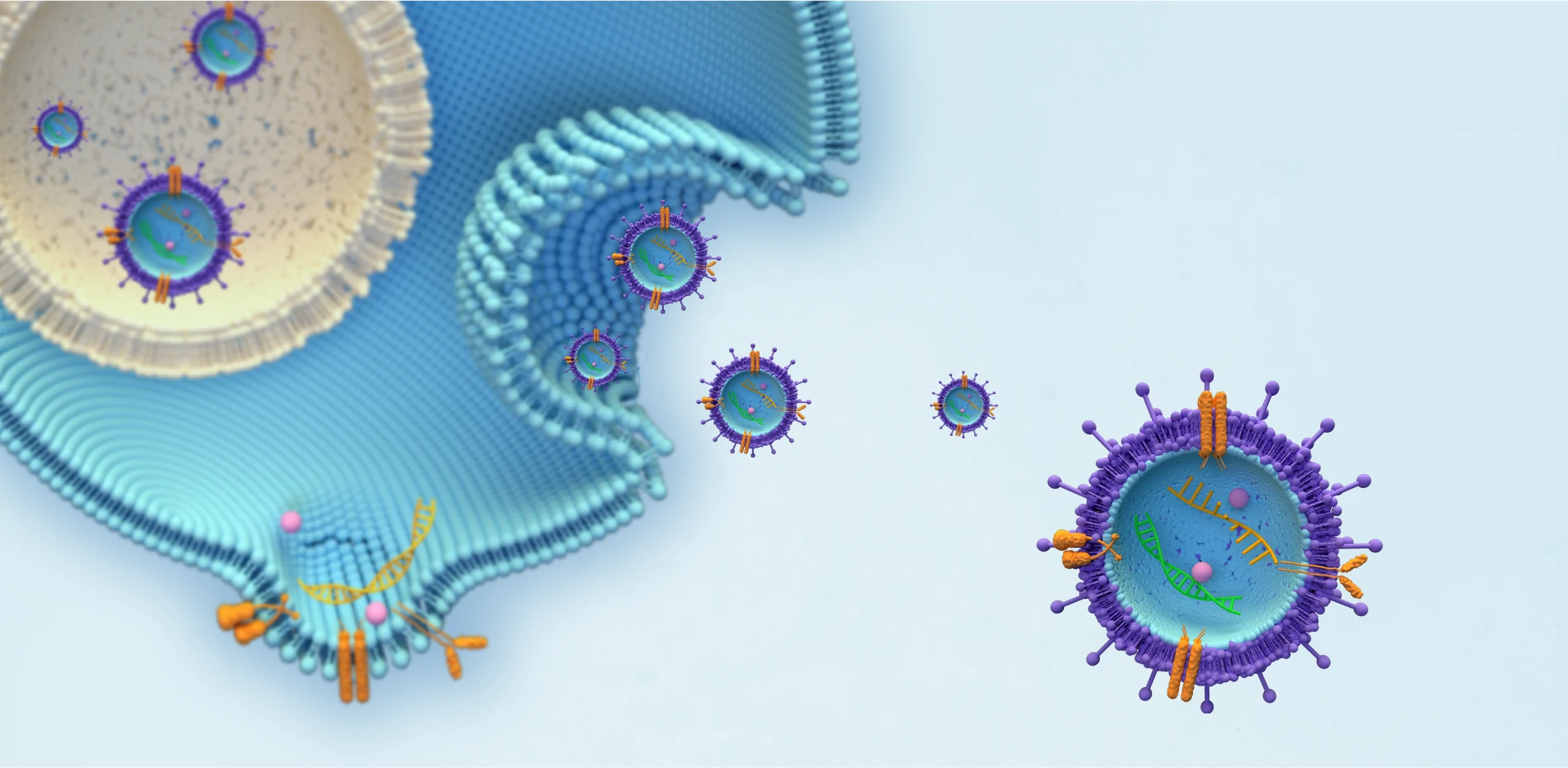
VIRUSES/VIRAL VACCINE

INTEGRATE INTO THE GENOMES OF THE HOST CELLS

OTHERS

COMPREHENSIVE MEASUREMENT SUITE FOR NANOPARTICLES AND INDIVIDUAL MITOCHONDRIA


EXTRACELLULAR VESICLES

THE ORIGIN AND PHENOTYPING OF EXTRACELLULAR VESICLES

VACCINE

COMPREHENSIVE MEASUREMENT SUITE FOR NANOSIZED DRUG PRODUCTS

NANOMEDICINE

COMPREHENSIVE MEASUREMENT SUITE FOR NANOSIZED DRUG PRODUCTS

BACTERIA DETECTION

TRULY QUANTITATIVE MEASUREMENT OF TOTAL BACTERIAL TITRE

VIRUSES/VIRAL VACCINE

INTEGRATE INTO THE GENOMES OF THE HOST CELLS

OTHERS

COMPREHENSIVE MEASUREMENT SUITE FOR NANOPARTICLES AND INDIVIDUAL MITOCHONDRIA

Advancing CAR-T Development With NanoFCM: Characterising Viral and Non-Viral Delivery Systems :
As CAR-T therapies continue to evolve, both viral and non-viral delivery systems are emerging as powerful platforms for nucleic acid delivery and cellular engineering. However, the successful development, optimisation, and translation of these complex delivery systems depends heavily on access to precise, high-resolution analytical technologies. In this webinar, we will demonstrate how nano-flow cytometry (NanoFCM) has become an essential tool for the characterisation of both viral vectors and non-viral nanoparticles, delivering critical single-particle insights that conventional techniques often cannot achieve. Attendees will learn how NanoFCM enables accurate measurement of particle size, concentration, purity, cargo encapsulation efficiency, and ligand quantification at the single-particle level. These capabilities support process control, enhanced reproducibility, and accelerated development of next-generation CAR-T therapies. Through real-world applications, we will showcase the growing role of NanoFCM as a foundational analytical platform for advanced cell and gene therapy workflows.
However, the successful development, optimisation, and translation of these complex delivery systems depends heavily on access to precise, high-resolution analytical technologies. In this webinar, we will demonstrate how nano-flow cytometry (NanoFCM) has become an essential tool for the characterisation of both viral vectors and non-viral nanoparticles, delivering critical single-particle insights that conventional techniques often cannot achieve.
Attendees will learn how NanoFCM enables accurate measurement of particle size, concentration, purity, cargo encapsulation efficiency, and ligand quantification at the single-particle level. These capabilities support process control, enhanced reproducibility, and accelerated development of next-generation CAR-T therapies.
Through real-world applications, we will showcase the growing role of NanoFCM as a foundational analytical platform for advanced cell and gene therapy workflows.
Date :
June 25, 2026
Time :
7:00 AM PT, 10:00 AM EDT, 4:00 PM CET
30 +
Countries
300 +
Top Users
80 %
TOP 20 Biopharma
1000 +
Publications
Loading more content...