
Disease Diagnosis
Exosomes exist in all bodily fluids. Due to their biological content and composition, they pose a perfect candidate for disease diagnosis, therapy and prognosis monitoring.
Exosomes exist in all bodily fluids. Due to their biological content and composition, they po...

Drug Delivery
Exosomes can be artificially loaded with proteins, nucleic acids, and small molecule drugs, etc., to effectively deliver their cargo, improve patient drug efficiency, reduce drug administration frequency, and reduce side effects of systemic exposure.
Exosomes can be artificially loaded with proteins, nucleic acids, and small molecule drugs, et...
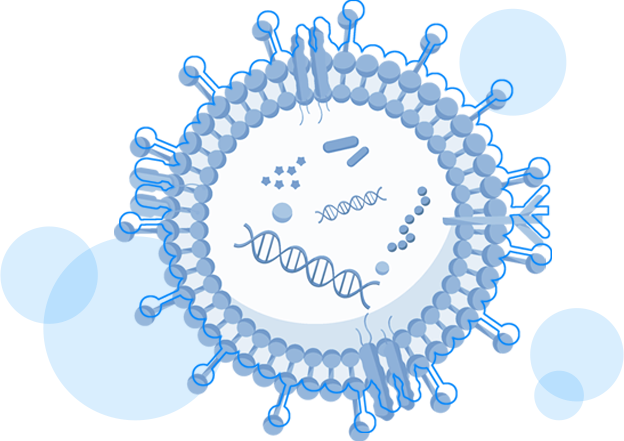
Exosomes from various sources
Small EVs (<200nm) are nanoparticles that carry a limited amount of cargo molecules such as proteins, nucleic acids and lipids. They can heterogeneous in size and in contents.
Small EVs (<200nm) are nanoparticles that carry a limited amount of cargo molecules such as...

Isolation and Characterization Methods
A wide variety of biological functions have been ascribed to Evs including immune response, antigen presentation, cellular differentiation and tumor invasion, all which can play a role in various diseases and contribute to their physiological processes. Isolation of EVs from complex biological fluids with high purity is essential for the accurate analysis of EVs and their cargo.
A wide variety of biological functions have been ascribed to Evs including immune response, an...

Stem cells exosomes
Exosomes derived from stem cells could affect cells regeneration and repair. They have a great potential in therapeutics attributed to their low immunogenicity.
Exosomes derived from stem cells could affect cells regeneration and repair. They have a great...




