Red blood cell-derived EVs in AML therapy
Author: admin Date: February 21, 2024
Acute Myeloid Leukaemia (AML) is the most common blood cancer in adults. Although 2 out of 3 AML patients go into total remission after chemotherapy and targeted therapies, the disease recurs in 60%-65% of younger adult patients within 3 years after diagnosis with a dramatically decreased survival rate. Therapeutic oligonucleotides are promising treatments under development for AML as they can be designed to silence oncogenes with high specificity and flexibility. However, validated approaches for safely and efficiently delivering oligonucleotide drugs are limited. This issue could be resolved by utilizing a new generation of delivery vehicles such as extracellular vesicles (EVs).
A platform to purify large-scale quantities of red blood cell-derived EVs (RBCEVs) (1013–1014 EVs from each blood unit) in a low cost and time-efficient fashion has been validated. In a previous study, a variety of RNA therapeutics including ASOs, gRNAs, and Cas9 mRNA were loaded into RBCEVs and subsequently delivered to target cells, showing significant therapeutic effects in, in vitro and in vivo models of breast cancer and AML. In this study, the authors harness RBCEVs and engineer them via exogenous drug loading and surface functionalization to develop an efficient drug delivery system for AML and characterise the single particles using the Flow NanoAnalyzer.
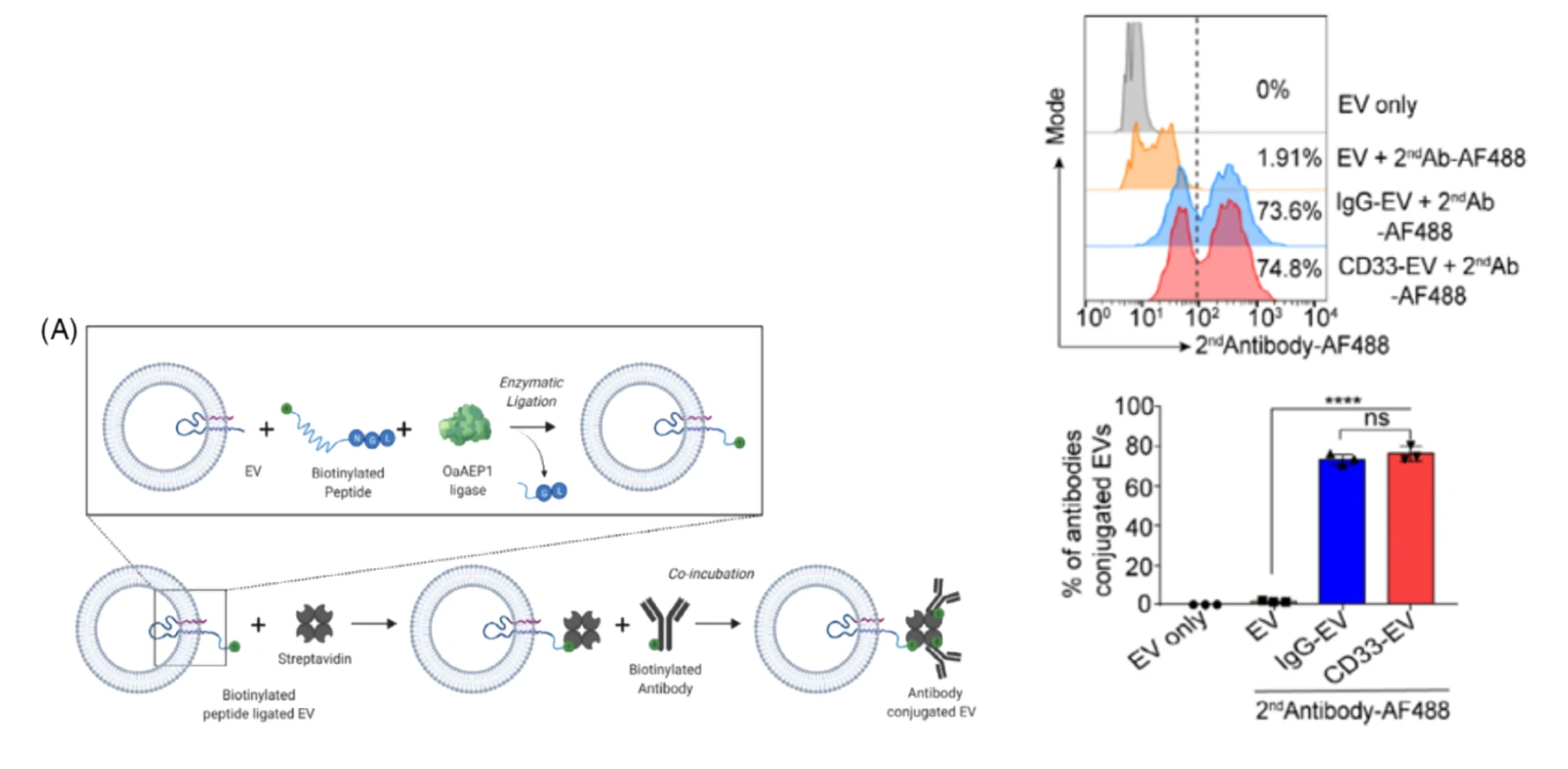
Figure 1. EVs surface antibody modification strategies Figure 2. The Flow NanoAnalyzer provides an estimation of the modification efficiency
To estimate the antibody conjugation efficiency at single-RBCEV level, the Flow NanoAnalyzer was used to detect RBCEVs coated with biotinylated anti-CD33 antibody and isotype monoclonal antibody (IgG-EV). The nano-flow cytometric analysis indicated that ~74% of RBCEVs were successfully conjugated with the CD33 monoclonal antibody. Subsequent in vivo experiments demonstrated that RBCEVs loaded with ASO significantly suppresses AML progression. Nano-flow cytometry plays a crucial role in the validation of exosome modification results and is expected to advance the development of engineered EVs drugs.
Cell Prolif., 2022, 55(9):e13255.